Effective atomic number
Effective atomic number has two different meanings: one that is the effective nuclear charge of an atom, and one that calculates the average atomic number for a compound or mixture of materials. Both are abbreviated Zeff.
For an atom
The effective atomic number Zeff, (sometimes referred to as the effective nuclear charge) of an atom is the number of protons an electron in the element effectively 'sees' due to screening by inner-shell electrons. It is a measure of the electrostatic interaction between the negatively charged electrons and positively charged protons in the atom. One can view the electrons in an atom as being 'stacked' by energy outside the nucleus; the lowest energy electrons (such as the 1s and 2s electrons) occupy the space closest to the nucleus, and electrons of higher energy are located further from the nucleus.
The binding energy of an electron, or the energy needed to remove the electron from the atom, is a function of the electrostatic interaction between the negatively charged electrons and the positively charged nucleus. In Iron, atomic number 26, for instance, the nucleus contains 26 protons. The electrons that are closest to the nucleus will 'see' nearly all of them. However, electrons further away are screened from the nucleus by other electrons in between, and feel less electrostatic interaction as a result. The 1s electron of Iron (the closest one to the nucleus) sees an effective atomic number (number of protons) of 25. The reason why it is not 26 is because some of the electrons in the atom end up repelling the others, giving a net lower electrostatic interaction with the nucleus. One way of envisioning this effect is to imagine the 1s electron sitting on one side of the 26 protons in the nucleus, with another electron sitting on the other side; each electron will feel less than the attractive force of 26 protons because the other electron contributes a repelling force. The 4s electrons in Iron, which are furthest from the nucleus, feel an effective atomic number of only 5.43 because of the 25 electrons in between it and the nucleus screening the charge.
Effective atomic numbers are useful not only in understanding why electrons further from the nucleus are so much more weakly bound than those closer to the nucleus, but also because they can tell us when to use simplified methods of calculating other properties and interactions. For instance, Lithium, atomic number 3, has two electrons in the 1s shell and one in the 2s shell. Because the two 1s electrons screen the protons to give an effective atomic number for the 2s electron close to 1, we can treat this 2s valence electron with a hydrogenic model.
Mathematically, the effective atomic number Zeff can be calculated using methods known as "self-consistent field" calculations, but in simplified situations is just taken as the atomic number minus the number of electrons between the nucleus and the electron being considered.
For a compound or mixture
Effective atomic number is a term that is similar to atomic number but is used for compounds (e.g. water) and mixtures of different materials (such as tissue and bone) rather than for atoms. Given some particular compound or mixture, the effective atomic number can in spirit be found by asking the question: What would the atomic number of a pure element have to be for the element to behave like this compound or mixture? The answer often involves a weighted average of the atomic numbers of the constituent atoms. One proposed formula for the effective atomic number, Zeff, is as follows:
![Z_{\text{eff}} = \sqrt[2.94]{f_{1} \times (Z_{1})^{2.94} %2B f_{2} \times (Z_{2})^{2.94} %2B f_{3} \times (Z_{3})^{2.94} %2B ...}](/2012-wikipedia_en_all_nopic_01_2012/I/efe25af14b6a9e88727a42c54d8a1711.png)
-
- where
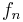 is the fraction of the total number of electrons associated with each element, and
is the fraction of the total number of electrons associated with each element, and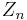 is the atomic number of each element.
is the atomic number of each element.
- where
-
An example is that of water (H2O), made up of two hydrogen atoms (Z=1) and one oxygen atom (Z=8), the total number of electrons is 1+1+8 = 10, so the fraction of electrons for the two hydrogens is (2/10) and for the one oxygen is (8/10). So the Zeff for water is:
Effective atomic number is important for predicting how X-rays interact with a substance, as certain types of x-ray interactions depend on the atomic number. The exact formula, as well as the exponent 2.94, can depend on the energy range being used.
This 'power law' method, while commonly employed, is of questionable appropriateness in contemporary scientific applications within the context of radiation interactions in heterogeneous media. This approach dates back to the late 1930s when photon sources were restricted to low-energy x-ray units (Mayneord 1937). The exponent of 2.94 relates to an empirical formula for the photoelectric process which incorporates a ‘constant’ of 2.64 x 10-26, which is in fact not a constant but rather a function of the photon energy. A linear relationship between Z2.94 has been shown for a limited number of compounds for low-energy x-rays, but within the same publication it is shown that many compounds do not lie on the same trendline (Spiers et al. 1946). As such, for polyenergetic photon sources (in particular, for applications such as radiotherapy), the effective atomic number varies significantly with energy (Taylor et al. 2008). As shown by Taylor et al. (2008), it is possible to obtain a much more accurate single-valued Zeff by weighting against the spectrum of the source. The effective atomic number for electron interactions may be calculated with a similar approach (Taylor et al. 2009).
References
- Webelements
- Eisberg and Resnick, Quantum Physics of Atoms, Molecules, Solids, Nuclei, and Particles.
- R. C. Murty, "Effective atomic numbers of heterogeneous materials", Nature 207, 398-399 (24 July 1965)
- W. Mayneord, “The significance of the Röntgen”, Unio Internationalis Contra Cancrum, 2, 271-282 (1937)
- W. Spiers, “Effective atomic number and energy absorption in tissues”, British Journal of Radiology, 19, 52-63 (1946)
- M. L. Taylor et al, "The effective atomic number of dosimetric gels", Australasian Physical & Engineering Sciences in Medicine, 31, 131-138 (2008)
- M. L. Taylor et al, "Electron Interaction with Gel Dosimeters: Effective Atomic Numbers for Collisional, Radiative and Total Interaction Processes", Radiation Research, 171, 123-126 (2009)
![Z_{\text{eff}} = \sqrt[2.94]{0.2 \times 1^{2.94} %2B 0.8 \times 8^{2.94}} = 7.42](/2012-wikipedia_en_all_nopic_01_2012/I/de6bf8834cd1eb3caf14cb35d208147c.png)